We’ve previously discussed many important aspects of tick paralysis treatment, including how to appropriately administer tick anti-serum (TAS), and effective management of respiratory disease. While it’s true that these are the two most important considerations in treatment of tick paralysis patients, there’s a wide variety of supportive care and management techniques that can also be utilised to maximise every patient’s chances of a smooth recovery.

In this article, we’ll focus on these additional therapies for tick paralysis supportive care. These range from mainstays of treatment for many patients like fluid therapy and proper nursing care, all the way through to additional medications like gastroprotectants, supplements, and anti-emetics that can assist patients through the added challenges of tick paralysis. So read ahead to find out how you can best support your tick paralysis patient with effective supportive care.
Fluid Therapy
The use of intravenous fluids in dogs with tick paralysis has been associated with reduced duration of hospitalisation. There has been a historical reluctance to use intravenous fluid therapy because of a perceived risk of pulmonary oedema due to the cardiac aspects of tick toxicity. Intravenous fluids should be utilised in the setting of an objective fluid plan individually tailored to each patient. All patients will be nil per os for at least 24 hours, so fluid therapy is indicated from admission to maintain hydration and account for ongoing losses.
Rates for Crystalloid Therapy
- Hartmanns or Plasmalyte 148 administered at maintenance rates, though each patient will need to be assessed individually (e.g., dehydration status)
- Supplements added to fluids – KCl may be added due to replenish GIT losses from vomiting/regurgitation if indicated by test results

All routine monitoring and precautions for intravenous fluid use should be observed. While fluid therapy can be useful and hasten patient recovery, particular care should be taken to avoid over-hydration in cats. This species seems most susceptible to pulmonary complications associated with fluid overload.
Gastroprotectants
The use of gastric protectants to reduce gastric acidity is controversial in human and veterinary medicine due to an association with nosocomial infections, particularly pneumonia. On the other hand, oesophageal strictures have been reported anecdotally (and seen at AES/VSS) after tick paralysis presumably due to reflux. The following medication may be administered at the clinician’s discretion and a reason for use recorded in the clinical notes:
- Esomeprazole 0.5-1mg/kg/hr IV q12hrs
Esomeprazole is a proton pump inhibitor and is more effective than ranitidine for increasing gastric pH. Full action may be delayed, and there is no pro-kinetic effect, so the general recommendation is that if anti-ulcer medications are to be used they should both be given for the first 48 hours then esomeprazole continued as a sole medication if needed.
These medications do little to prevent pooling of oropharyngeal secretions in the oesophagus and their periodic emptying, seen most commonly as regurgitation – instead they reduce the chances of acidic stomach contents entering the oesophagus, which are potentially more damaging to both the oesophagus and the pulmonary tissue. Oesophageal/oral suctioning may help remove these secretions, however this can be painful
and requires heavy sedation if it is not to be detrimental to the patient.
Patient positioning can also help prevent aspiration of regurgitated material by keeping them in sternal recumbency with the trachea angled ventrally. Some patients may also tolerate placement of a naso-oesophageal feeding tube for regular decompression. NG tube placement is ideally avoided, to prevent reflux of stomach acid into the oesophagus. Use is limited by patient size as the tube needs to be greater than 6Fr diameter to allow effective removal of thick secretions.
Anti-Emetics
The rationale for anti-emetic use is to reduce the incidence of vomiting in order to ideally reduce the incidence of aspiration pneumonia. Patients presenting with a history of vomiting or regurgitation should be started on an antiemetic. The following anti-emetics can be used at the clinician’s discretion:
- Metoclopramide 0.5mg/kg IV, then CRI at 2mg/kg/day (C2)
- Maropitant 1mg/kg SC q24hrs (C2)
- Ondansetron 0.1-0.4mg/kg IV q8hrs (C2)
Metoclopramide may be a useful antiemetic because it increases gastric emptying and increases lower oesophageal sphincter tone, preventing reflux as well as acting as a central emetic.
Antibiotics
As discussed in our previous article on respiratory disease as a major component of tick paralysis, management of aspiration pneumonia is crucial. In patients that are suspected to have aspirated, or shown signs of aspiration pneumonia based on radiographic findings, commencement of antibiotic therapy is necessary. The following antibiotics are most commonly used in these situations:
- Ampicillin 22mg/kg IV q8hrs or Cephazolin 22mg/kg IV q8hrs
- Enrofloxacin 5mg/kg/hr SC q12hrs
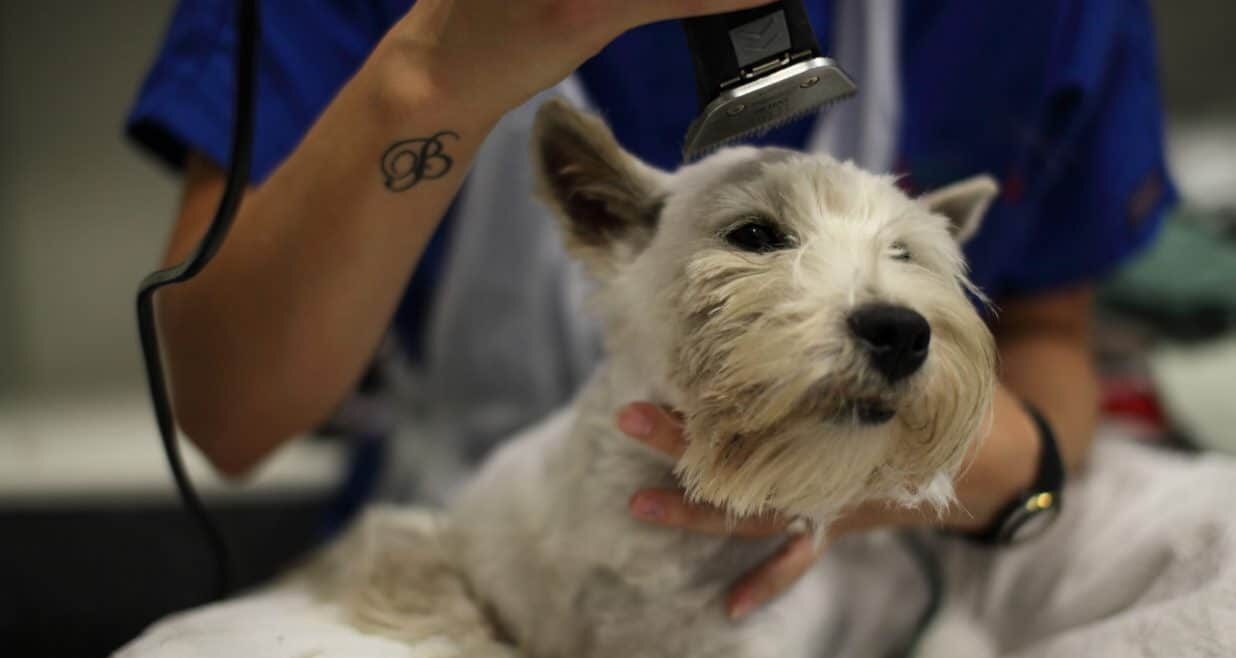
Entire Body Clipping
This is recommended highly in all cases of tick paralysis. In a recent study, 9% of cats with tick paralysis had a second tick identified. To prevent the continuing exposure to tick venom and ensuing paralysis requires removal of all ticks. This can be performed under sedation (mainly for dogs), and general anaesthesia (for all cats) to reduce stress and control respiration.
It is essential that all patients admitted with tick paralysis should receive a tick clip and tick bath as well as repeated tick searches at every check, as it is not uncommon to find more than one tick. This is particularly important, as attached ticks that were previously undetected may become more easily noticeable as they continue to feed and grow in size.
Nursing Care
The majority of tick paralysis patients are unable to self-position, urinate or even blink, so intensive nursing care is critical to reduce complications such as bladder atony or corneal ulceration, as well as improving patient comfort and recovery in hospital.
Soft, dry bedding and regular turning is important to prevent pressure sores. AES uses absorbent disposable bedding to reduce the amount of washing of sheets/towels, but once a patient urinates or defecates on its bedding it is changed immediately to reduce urine/faecal scalding. Regular bladder checks are indicated, with expression, or placement of an indwelling urinary catheter, to prevent detrusor muscle atony, overfill incontinence or urinary tract infections.
Regular artificial tear administration (every 2 hours) is required, especially if the tick was attached around the eye or CN5. The artificial tear product should be preservative-free, as preservatives in these preparations have been shown to induce corneal ulceration in humans (e.g Celluvisc).

Corneal ulceration secondary to desiccation needs to be identified quickly and can be treated with antibiotics and a temporary tarsorrhaphy (either non-surgical or surgical). As an alternative, AES now has access to contact lenses and has had success with preventing corneal ulceration for anaesthetised patients.
It is recommended that nursing staff perform a full TPR on tick patients every 2-4 hours. This includes SpO2 monitoring, bladder palpation and expression, application of artificial tears, oral moistening and patient turning. If the patient is receiving oxygen therapy or there are concerns about ventilation then oxygen saturations are measured every 2 hours and blood gas parameters are measured at the vet’s discretion.
The nursing and care of a patient being ventilated is beyond the scope of this protocol. Patients that are regurgitating and retching also require more intensive nursing care, as aspiration is a high risk especially in patients that are severely affected. Immediate suctioning of the oral and pharyngeal areas after a regurgitation event is of extreme importance to reduce the risk aspiration.
Discharge
Animals can be discharged when they are no longer ataxic and when eating and drinking can be accomplished without gagging, and voluntary urination documented. Water and food trials are usually implemented when the patient is walking well, has stopped regurgitating and has a palpable swallow or gag reflex. Monitoring during the trial is imperative and if there is any evidence of laryngeal dysfunction (as evidenced by a cough or retch) then a repeat trial is performed in 6-12 hours time.
Post discharge instructions need to include restricting activity, keeping the patient cool and supervision of eating and drinking until complete normality returns and monitoring of urination patterns.
Successful management of the tick paralysis case is multifactorial, and requires contribution from all members of the veterinary team to achieve a favourable patient outcome. Ensuring all staff are communicating effectively, records of patient monitoring and medication are transparent across the team, and adequate supportive care is provided frequently, can lead to a smooth and satisfying patient recovery.
Want to access the tick paralysis protocol in full? Sign up to VetAPedia for free access to 100+ clinical resources.